The EUROX® measurement system for extractive measurements on tin baths can detect smallest amounts of oxygen and determine several characteristic values like the dew point temperature. The gas to be investigated is heated up and catalytically decomposed. After reaction the oxygen content is measured at equilibrium and converted.
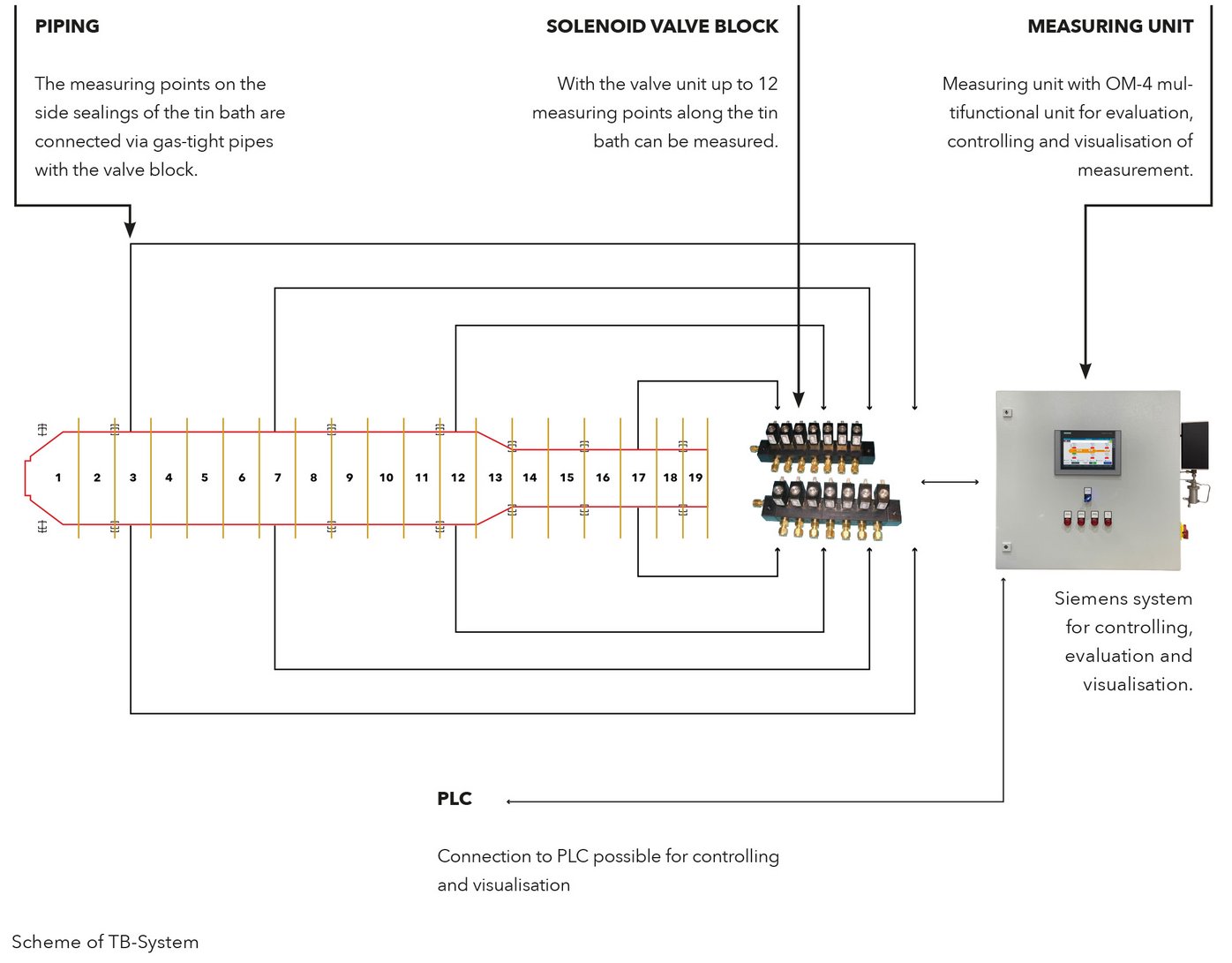
EUROX® TB-System includes a solenoid valve unit for switching between the connected measuring points and a measuring unit including an OM-4 multifunctional unit for evaluation, controlling and visualisation. The measuring system can be connected with the PLC, so controlling and visualisation can also be done via PLC.
Oxygen partial pressure measuring in tin bath
Also a HORN created key figure of the atmosphere based on the tin, oxygen and tin oxide is defined. This gives the controller a quick and scientifically sound statement about the overall state of the atmosphere and its effect on the tin bath.
Oxidation-State
The oxygen partial pressure in the tin bath atmosphere is measured and related to temperature dependent tin/tin-oxide equilibrium curves. This indicates the degree of oxidation of the tin bath surface.
The oxidation – state is defined as 1 on the tetravalent tin / tin oxide equilibrium curve. When the measured oxygen partial pressure is higher than the tetravalent equilibrium curve, then also the oxidation state is higher than 1.
Hydro-State
Hydrogen in the atmosphere does not only reduce the oxygen in the atmosphere, it can also reduce tin oxide to tin and water. The quotient water to hydrogen is a measure of the potential of this reaction.
The water content in the atmosphere can be calculated and related to the H 2 content. So the Hydro
– State is defined. This indicates a reasonable range of the H 2 content, but also gives an indication about the water content in the tin bath and thus also about the dissolved water content in the liquid tin.
If the measured quotient is equal to the temperature dependent maximum, the state is by definition equal to one. If it is less, then it is less than 1.
Comparison
All three characteristic values are compared according to their measurement location. If the dew point at the beginning of the tin bath indicates an air ingress, the oxidation - state shows that no significant oxidation of the tin bath takes place due to the high temperature in this area.
At the other end of the tin bath, however, the degree of oxidation is increased, which is mainly due to the low temperature.
However, the fact that the hydro-state is far below 1 in the entire tin bath shows that the protection of the liquid tin is given and could even be reduced somewhat, which would have a positive effect on the operating costs.
To avoid oxidation of the liquid tin in tin baths of float plants what would cause glass defects it is important to analyse and control the atmosphere in the tin bath. With the EUROX® TB-system up to 8 selected measuring points per measuring system along the tin bath can be monitored with just one sensor.
EUROX® sensors measure even at very low oxygen partial pressure. The gas to be investigated is heated up and catalytically decomposed. After reaction the oxygen content is measured at equilibrium and converted.
COOKIES
We use cookies to make it easier to use and to further improve our service.
If you have given us your consent, you can revoke it at any time in the data protection declaration.
COOKIES
Necessary
Necessary cookies help make a website usable by enabling basic functions such as page navigation and access to secure areas of the website. The website cannot function properly without these cookies.
C3 Cookie
| Name | Provider | Purpose | Expiration | Type |
|---|---|---|---|---|
| c3kie | hornglass.com | Saves the consent status of the user whether the cookie window should be displayed. | 180 days | HTTP Cookie |
Technical
Technichal cookies help to improve the user experience of the website. New functionalities will be enabled.
YouTube
| Name | Provider | Purpose | Expiration | Type |
|---|---|---|---|---|
| yt-player-headers-readable | YouTube | Used to determine the optimal video quality based on the visitor's device and network settings. | Persistent | HTML Local Storage |
| VISITOR_INFO1_LIVE | YouTube | Tries to estimate the range of users on pages with built-in YouTube videos. | 179 days | HTTP Cookie |
| YSC | YouTube | Registers a unique ID to keep statistics on which videos from YouTube the user has seen. | Session | HTTP Cookie |
| yt.innertube::nextId | YouTube | Registers a unique ID to keep statistics on which videos from YouTube the user has seen. | Persistent | HTML Local Storage |
| yt.innertube::requests | YouTube | Registers a unique ID to keep statistics on which videos from YouTube the user has seen. | Persistent | HTML Local Storage |
| ytidb::LAST_RESULT_ENTRY_KEY | YouTube | Saves the user's video player settings with embedded YouTube video. | Persistent | HTML Local Storage |
| yt-remote-cast-available | YouTube | Saves the user's video player settings with embedded YouTube video. | Session | HTML Local Storage |
| yt-remote-cast-installed | YouTube | Saves the user's video player settings with embedded YouTube video. | Session | HTML Local Storage |
| yt-remote-connected-devices | YouTube | Saves the user's video player settings with embedded YouTube video. | Persistent | HTML Local Storage |
| yt-remote-device-id | YouTube | Saves the user's video player settings with embedded YouTube video. | Persistent | HTML Local Storage |
| yt-remote-fast-check-period | YouTube | Saves the user's video player settings with embedded YouTube video. | Session | HTML Local Storage |
| yt-remote-session-app | YouTube | Saves the user's video player settings with embedded YouTube video. | Session | HTML Local Storage |
| yt-remote-session-name | YouTube | Saves the user's video player settings with embedded YouTube video. | Session | HTML Local Storage |
Advertisement
Advertisement cookies allow our advertising partners to show you ads that fit your interests.
TikTok
| Name | Provider | Purpose | Expiration | Type |
|---|---|---|---|---|
| _ttp | TikTok | Used to store a unique user ID. | 1 year | HTTP Cookie |
| Name | Provider | Purpose | Expiration | Type |
|---|---|---|---|---|
| _fbp | Used to store and track visits to websites. | 3 months | HTTP Cookie |
Statistics
Statistics cookies help website owners understand how visitors interact with websites by collecting and reporting information anonymously.
Google Tag Manager
| Name | Provider | Purpose | Expiration | Type |
|---|---|---|---|---|
| _ga | Google Tag Manager | Registers a unique ID that is used to generate statistical data on how the visitor uses the website. | 2 years | HTTP Cookie |
| _ga_ | Google Tag Manager | Collects data on how often a user visited a website, as well as data on the first and last visit. Used by Google Analytics. | 2 years | HTTP Cookie |
Microsoft Clarity
| Name | Provider | Purpose | Expiration | Type |
|---|---|---|---|---|
| _clck | Microsoft | Persists the Clarity User ID and preferences, unique to that site is attributed to the same user ID. | 1 year | HTTP Cookie |
| _clsk | Microsoft | Connects multiple page views by a user into a single Clarity session recording. | 1 year | HTTP Cookie |
| CLID | Microsoft | Identifies the first-time Clarity saw this user on any site using Clarity. | 1 year | HTTP Cookie |
| ANONCHK | Microsoft | Indicates whether MUID is transferred to ANID, a cookie used for advertising. Clarity doesn't use ANID and so this is always set to 0. | 1 year | HTTP Cookie |
| MR | Microsoft | Indicates whether to refresh MUID. | 1 year | HTTP Cookie |
| MUID | Microsoft | Identifies unique web browsers visiting Microsoft sites. These cookies are used for advertising, site analytics, and other operational purposes. | 1 year | HTTP Cookie |
| SM | Microsoft | Used in synchronizing the MUID across Microsoft domains. | 1 year | HTTP Cookie |